Extensive loss of muscle mass known as muscle atrophy is a devastating complication of a large number of chronic disease states such as cancer, diabetes, chronic heart, pulmonary and renal diseases and of neurodegenerative diseases. Muscle atrophy, also known as Sarcopenia occurs also as the natural process of ageing which cause low mobility and accelerated mortality. No treatment is presently available to this multi-disease complication.
Our current interest is in the elucidation of signaling molecules that are involved in muscle atrophy. Recently, we found that p38α mitogen-activated protein kinase (MAPK) is a key intracellular signaling pathway that coordinates the activities of several metabolic pathways that together induce atrophy of denervated muscles. P38α MAPK is involved in the accumulation of mitochondrial damage and oxidative stress. Its activity blocks removal of damaged mitochondria by autophagy and enhances protein degradation by the ubiquitin-proteasome system. Therefore, the p38α MAPK pathway is a promising target for preventing muscle atrophy and weakness in many diseases (Odeh et al 2019).
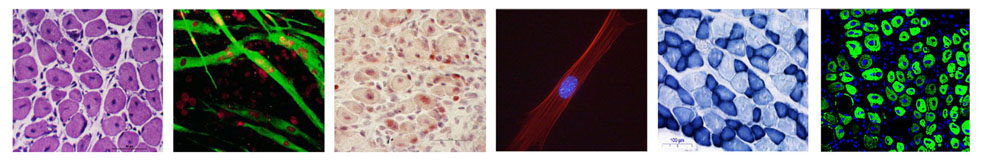
Myofiber with satellite cells
Ongoing and planned projects:
1. Inhibition of p38 MAPK in several disease models that involve muscle atrophy.
We will investigate mouse models of Diabetes and of Amyotrophic Lateral Sclerosis (ALS). The activity of p38 MAPK will be inhibited by genetic and by pharmacological means in muscles of well–established mouse models of Diabetes and ALS. Skeletal muscle metabolism will be elucidated using advanced metabolomics analyses. We anticipate to better understand how p38 MAPK affect muscle catabolism in these diseases and expect that the results of this research will add p38 MAPK as a potential target for prevention of muscle atrophy in disease states.
2. Elucidation of muscle-secreted cytokines that activate p38α MAPK pathway and myofiber atrophy in denervated muscles.
In collaboration with Prof. Lorenzo Puri (Sanford-Burnham Medical Research Institute, La Jolla, USA), we perform screening analyses to identify muscle-secreted cytokines that activate the p38α MAPK pathway in myofibers. In this study, muscles are denervated and the processes involved in muscle atrophy are being investigated. The study combines cell culture (in vitro) and animal (in vivo) studies towards the identification of muscle-secreted ligands activating p38α MAPK. We expect that knowledge of upstream activators of p38α MAPK which facilitates muscle atrophy will provide a more complete understanding of the pathways that initiate this process in many diseases, and will add to the repertoire of potential therapeutic targets in neurodegenerative diseases that involve loss of neuro muscular junction.

Electron micrograph of a longitudinal section of sarcomeres